 
What is the Difference Between Quicklime and Lime Water? This reaction is strongly exothermic and takes place vigorously with the formation of clouds of steam. When water is added to lime calcium hydroxide Ca(OH)2 is formed according to the following reaction. The formula for lime water is Ca(OH)2 and the chemical name for lime water is calcium hydroxide. 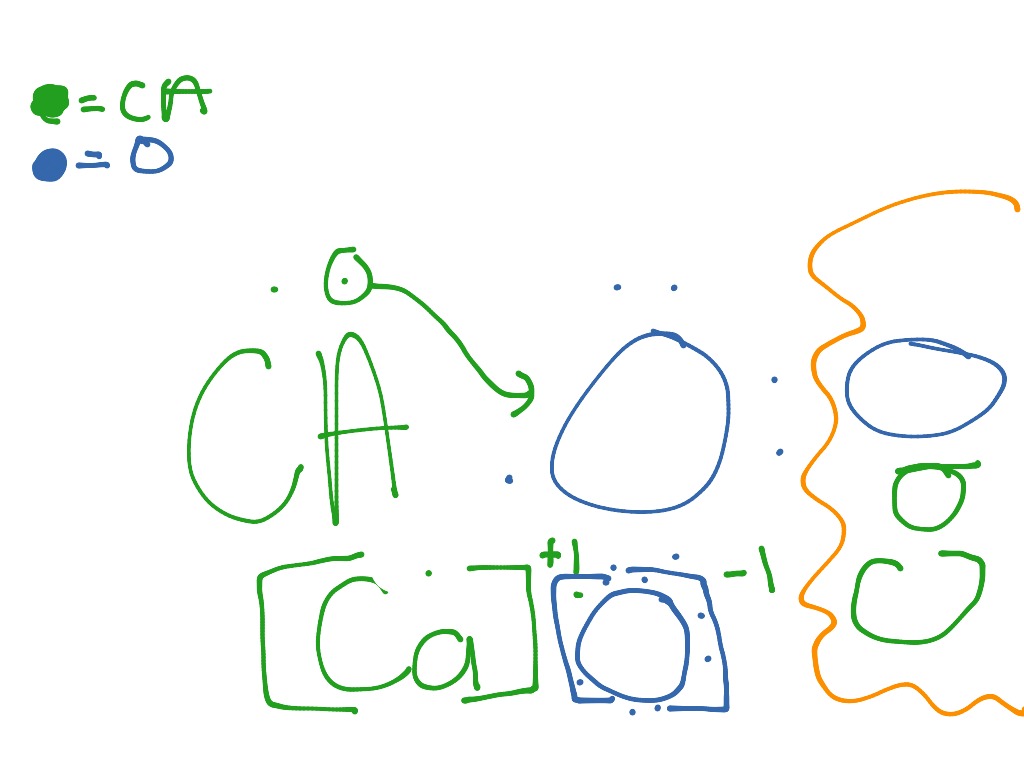
Thus, it can be understood that calcium oxide is an ionic compound featuring an ionic bond between calcium and oxygen. The structure of calcium oxide is illustrated below. This reaction is reversible and exothermic in nature in the forward direction.Ĭalcium oxide molecules contain one calcium cation (which holds a charge of +2) and one oxygen anion (which holds a charge of -2). The carbon dioxide that is formed is immediately removed so that the reaction is preceded until the completion of the process in accordance with Le-Chatelier’s principle. The products formed as a result of the reaction are burnt lime and carbon dioxide. These reactions are usually held in a rotary kiln. The process that is used to prepare burnt lime is known as calcination.Ĭalcium oxide is a process that starts with thermally decomposing the reactants at high temperatures but ensuring that the temperature is kept well below the melting point.Ĭalcium carbonate undergoes calcination at temperatures ranging between 1070oC-1270oC. This material isn’t volatile at ceramic temperatures.Ĭalcium oxide has a moderate effect on colour, except in large amounts when it may have a bleaching effect on iron oxide.Ĭalcium oxide also exists in the colour of kaki/tomato reds.Ĭalcium oxide can be produced by thermal decomposition of materials like limestone or seashells that contain calcium carbonate (CaCO3, mineral calcite) in a lime kiln. Calcium oxide, commonly known as lime, is a chemical compound with the formula CaO.Ĭalcium oxide, also known as quicklime, is an alkaline substance that has been in use since the medieval age.Ĭalcium oxide is believed that quicklime is one of the oldest chemicals known to the human race.Ĭalcium oxide can also be referred to as burnt lime or lime.Ĭalcium oxide has a medium viscosity and a high surface tension, plus a high to intermediate expansion and contraction rate. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
